Regulatory milestone
Nexstim Plc has received U.S. Food and Drug Administration clearance to market its NBS System 6 for the adjunct treatment of adult patients with Obsessive–Compulsive Disorder (OCD). The decision adds OCD to the system's existing U.S. therapy indication for major depressive disorder (MDD), and the NBS 6 remains FDA-cleared for pre-surgical mapping of speech and motor cortices.
Device and platform features
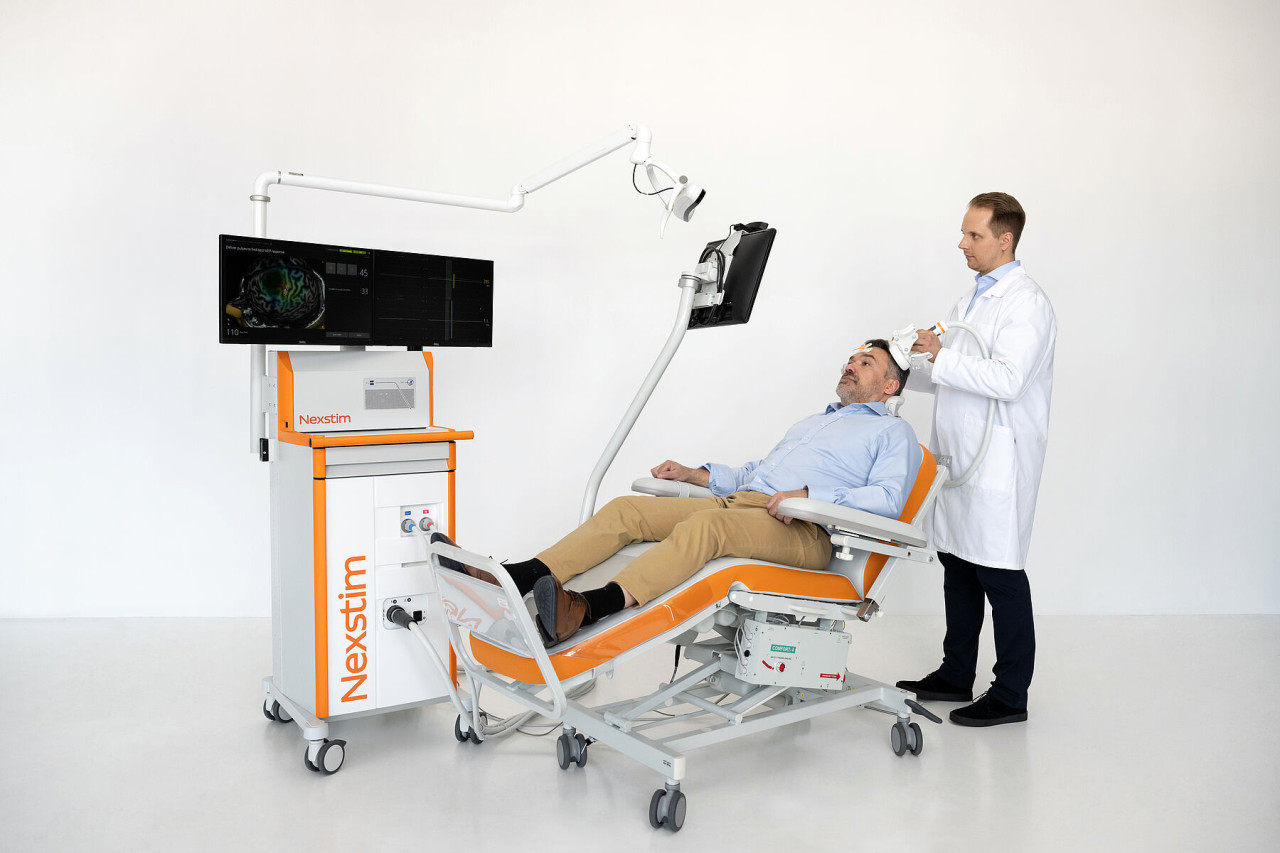
The NBS System 6 is Nexstim's latest generation of electric-field (E-field) navigated transcranial magnetic stimulation (nTMS). The platform couples subject-specific 3D navigation — using each patient's MRI anatomy — with real-time E-field estimation to target stimulation precisely to cortical sites. The company highlights a modular hardware design and a software-guided application workflow intended to simplify clinical use and allow expansion of capabilities for research or clinic-specific protocols.
Platform highlights:
- Navigation: MRI-based 3D navigation with E-field modelling enables individualized cortical targeting.
- Usability: a software-guided treatment application streamlines setup and session delivery for clinicians.
- Modularity: hardware and software modules can be added or customized over time to support different diagnostic or therapeutic functions.
Indications and clinical context
The NBS 6 is cleared in the U.S. for: adjunctive therapy for adult OCD, treatment of adults with MDD who have not responded adequately to antidepressant medication in the current episode, and pre-surgical mapping of speech and motor function.
Clinical evidence supporting TMS in OCD includes randomized trials such as Carmi et al. (American Journal of Psychiatry, 2019), which demonstrated symptom reduction with targeted stimulation paradigms. Nexstim's clearance indicates the FDA evaluated device-specific data and procedures sufficient to expand its therapy labeling to include OCD as an adjunctive treatment; the company previously announced therapy-focused availability in 2023 and diagnostic availability in October 2025.
Implications for clinicians and patients
The FDA clearance widens clinician options for non-invasive neuromodulation in treatment-resistant or adjunctive care for OCD. Practical implications include:
- Integration into care pathways: the NBS 6 can be considered as an adjunct to pharmacotherapy and psychotherapy for adult patients who remain symptomatic.
- Precision targeting: E-field navigation may help clinicians deliver stimulation to individualized cortical targets associated with symptom improvement, which could support more consistent treatment dosing across patients.
- Facility needs: adoption will require MRI-based workflows, staff training on navigated TMS protocols, and infrastructure for multi-session outpatient delivery.
Market and competitive landscape
The clearance positions Nexstim's NBS 6 within a competitive TMS market that includes several platforms offering different stimulation modalities and targeting approaches. For health systems and outpatient clinics, device selection will depend on clinical evidence, workflow, cost, reimbursement, and preferences for navigated versus non-navigated or deep TMS systems.
Company statement
Nexstim CEO Mikko Karvinen said the company is "pleased to announce another important addition to our therapy indication catalogue," and emphasized the company's mission to expand patient access to personalized non-invasive brain diagnostics and therapies.
Takeaways
Nexstim's NBS System 6 is now FDA-cleared as an adjunct treatment for adult OCD and continues to be cleared for MDD therapy and pre-surgical cortical mapping. The platform's E-field navigated approach and modular design aim to deliver personalized targeting and a flexible clinical workflow, potentially broadening non-invasive neuromodulation options for clinicians treating OCD and related disorders.
About Nexstim
Nexstim is a Finland-based medical technology company that develops navigated transcranial magnetic stimulation systems for diagnostics and therapy. The company markets the NBS System 6 for both pre-surgical mapping of cortical function and therapeutic applications. Nexstim's shares trade on Nasdaq First North Growth Market Finland.
References
Carmi L., et al. Targeted transcranial magnetic stimulation for obsessive–compulsive disorder. American Journal of Psychiatry. 2019;176:931–938.
Photo credit: www.nexstim.com
Tags: Nexstim, navigated TMS, OCD, FDA clearance, NBS 6
Topics: Non-invasive brain stimulation, Neuromodulation, Mental health technology