Executive appointment and funding
Avation Medical announced on March 20, 2026 that Manish Vaishya, PhD has been appointed Chief Executive Officer concurrent with the closing of a Series D financing round. The company said the new funding and leadership will accelerate commercialization, reimbursement strategy, pipeline expansion and international market entry for its Vivally System, a prescription wearable neuromodulation and digital health platform for overactive bladder (OAB) symptoms.
Background on the new CEO
Dr. Vaishya co-founded Avation and has served as Chief Technology Officer since the company’s inception. In that role he led development of the Vivally System through multiple clinical trials, achieved ISO 13485 quality certification, secured FDA regulatory clearance for the device, and supported the company’s U.S. market launch. Avation describes him as a MedTech executive with more than 25 years of experience developing and commercializing Class II and III medical devices and healthcare systems. His promotion to CEO formalizes leadership continuity as Avation moves from clinical development into broader commercial scale-up.
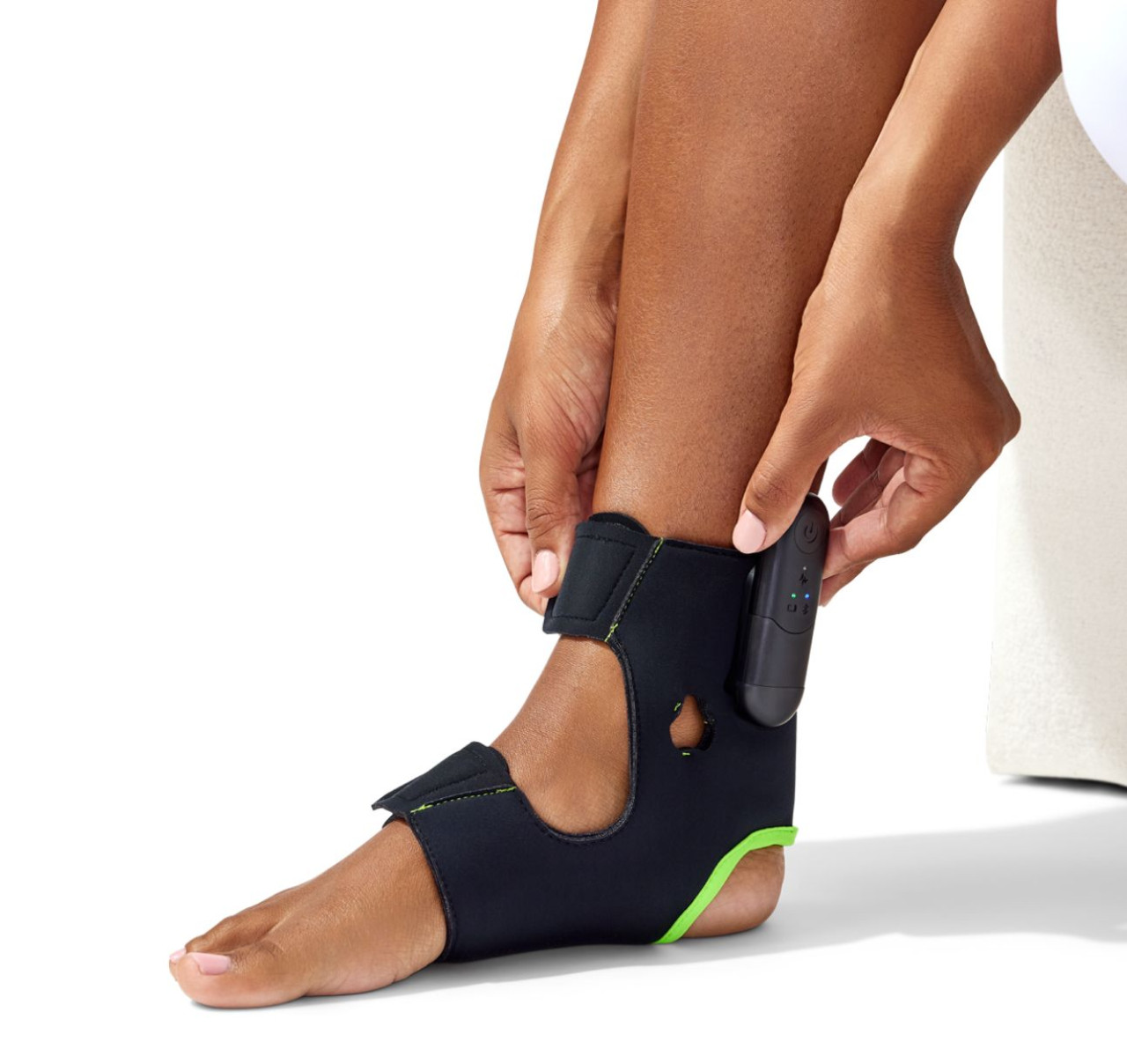
About the Vivally System
The Vivally System is an at-home, wearable neuromodulation therapy combined with a mobile app and eDiary. Avation markets Vivally as the only FDA-cleared physiologic closed-loop, prescription wearable device for treating symptoms of overactive bladder, including urge urinary incontinence and urinary urgency. According to the company, Vivally delivers therapy in 30-minute sessions and uses closed-loop control to tailor stimulation based on physiologic signals and patient response. Vivally is positioned as a drug-free, non-surgical alternative for patients who seek neuromodulation delivered outside clinical settings.
Commercial and strategic priorities
Avation said the Series D proceeds will be directed to multiple priorities intended to support broader patient access and company growth:
- Accelerating U.S. commercialization and building direct and channel sales infrastructure.
- Advancing reimbursement strategy, payer engagement and real-world evidence generation to support coverage.
- Expanding the product pipeline and development programs for additional clinical indications or geographies.
- Scaling manufacturing, quality systems and international market entry efforts.
Regulatory and market context
The announcement reiterates that Vivally is available in the United States by prescription following FDA clearance. The device’s positioning—wearable, closed-loop, and at-home—reflects a growing trend in neuromodulation toward patient-managed therapies and digital-health integrations. For clinicians and payers, the principal evidence considerations remain randomized trial outcomes, real-world adherence, safety in long-term use, and payer economics. Avation’s emphasis on reimbursement and real-world data generation aligns with the broader industry requirement to pair technological innovation with robust cost-effectiveness and outcomes evidence.
Outlook
With Dr. Vaishya as CEO, Avation is signaling continuity in product leadership while prioritizing commercialization and market access. The Series D financing provides capital to scale operations and pursue broader uptake of a non-invasive neuromodulation option for patients with overactive bladder. Success will depend on execution across sales, manufacturing, clinical evidence and payer engagement as the company seeks to extend access to the hundreds of millions of patients worldwide who experience urinary urgency and incontinence.
For more information
Vivally is distributed by prescription in the U.S.; Avation lists additional company information and clinician resources on its website. The company is headquartered in Columbus, Ohio.
Photo credit: www.avation.com
Tags: neuromodulation, wearable neurotech, overactive bladder, Vivally, Series D
Topics: Neurotech industry & startups, Neuromodulation, Wearable neurotech